Rural Prosperity and India’s Pharmaceuticals in Global Healthcare
S. Ahmad
In the architecture of modern economies, health is not merely a social good—it is a foundational economic asset. A healthy population drives productivity, innovation, and social stability, while a strong healthcare system reduces inequality and vulnerability. At the heart of this intersection between public health and economic progress stands the pharmaceutical industry. In India, this sector has evolved into far more than a manufacturing domain; it has become a strategic pillar of national development and a vital contributor to global healthcare equity.
Health functions both as a driver and an outcome of economic development, and the pharmaceutical industry occupies a unique position at this intersection. By ensuring the availability of essential medicines and vaccines, the industry plays a critical role in advancing public health outcomes and societal welfare, while concurrently generating employment, reinforcing supply-chain resilience, and contributing to broader socio-economic development. Over time, the Indian pharmaceutical industry has emerged as one of the world’s most distinctive and socially significant sectors, positioning India among the largest and most technologically advanced pharmaceutical producers globally.
Over the past few decades, India’s pharmaceutical industry has transitioned from domestic self-sufficiency to global leadership. It is now widely known as the “Pharmacy of the World,” a title earned through a rare combination of affordability, scale, and regulatory reliability. The industry has not only ensured the availability of essential medicines and vaccines for millions at home but has also supplied life-saving drugs to more than 190 countries. In doing so, it has linked rural manufacturing hubs and research laboratories to international markets, creating employment, strengthening supply chains, and enhancing India’s global standing.

India in the Global Pharmaceutical Landscape
India currently ranks third globally in pharmaceutical production by volume and eleventh by value. With over 3,000 pharmaceutical companies and approximately 10,500 manufacturing units, the country possesses one of the largest and most diversified drug production ecosystems in the world. The domestic pharmaceutical market, presently valued at around USD 60 billion, is projected to reach USD 130 billion by 2030—a reflection of expanding healthcare access, rising incomes, and growing global demand.
India’s standing as the Pharmacy of the World is rooted in the unique combination of affordable pricing and assured quality, which has made Indian medicines widely preferred across global markets. Cost-efficient manufacturing, supported by a strong scientific workforce and scale, has enabled the consistent supply of essential drugs without compromising standards.
According to the Economic Survey 2025–26, the sector’s annual turnover in FY25 reached ₹4.72 lakh crore, with exports growing at a compound annual growth rate of 7 percent over the past decade. Pharmaceutical exports in 2024–25 stood at USD 30.5 billion—an almost sixteen-fold rise from USD 1.9 billion in 2000–01. These figures underscore not only commercial growth but also deep integration into global healthcare supply chains.
India accounts for nearly 20 percent of the global supply of generic medicines and manufactures around 60,000 generic brands across 60 therapeutic categories. Its contribution has been particularly transformative in the fight against HIV/AIDS, tuberculosis, malaria, and other infectious diseases, where affordable generics have dramatically expanded treatment access across developing nations.
One of the strongest indicators of international trust lies in regulatory compliance. India hosts the highest number of manufacturing plants approved by the United States Food and Drug Administration (USFDA) outside the United States. This regulatory endorsement reflects adherence to stringent quality standards and reinforces global confidence in Indian pharmaceuticals. Additionally, the country is home to approximately 500 active pharmaceutical ingredient (API) manufacturers, representing nearly 8 percent of the global API industry.
Vaccine Leadership and Global Health Equity
India’s pharmaceutical strength extends beyond generics into vaccine manufacturing. Indian firms are global leaders in the supply of Diphtheria, Tetanus, and Pertussis (DPT), Bacillus Calmette–Guérin (BCG), and measles vaccines. About 60 percent of vaccine supplies procured by UNICEF are sourced from India, while 40–70 percent of global demand for DPT and BCG vaccines is met by Indian manufacturers. Remarkably, 90 percent of the measles vaccine demand of the World Health Organization (WHO) is supplied by Indian companies.
This dominance is not merely commercial success—it is a cornerstone of global public health. Affordable vaccines manufactured in India form the backbone of immunisation drives across Africa, Asia, and Latin America, directly saving millions of lives annually.
Expanding Export Horizons
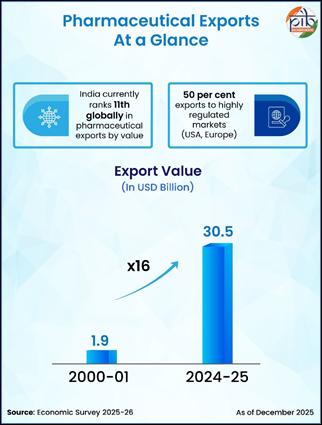
India’s pharmaceutical exports now reach 191 countries, with nearly half directed toward highly regulated markets such as the United States and Europe. This distribution pattern demonstrates not only geographical diversification but also acceptance in markets with the world’s strictest quality requirements.
Export momentum has remained robust. Monthly exports of drugs and pharmaceuticals rose from USD 2.59 billion in January 2025 to USD 2.66 billion in January 2026. Meanwhile, medical device exports increased from USD 2.5 billion in 2020–21 to USD 4.1 billion in 2024–25, with shipments to 187 countries.
Indian pharmaceutical exporters have also diversified into emerging and non-traditional markets, including Nigeria, Mexico, the United Republic of Tanzania, Brazil, Sri Lanka, Saudi Arabia, and Spain. By broadening their export portfolio across bulk drugs, formulations, and surgical products, companies have reduced exposure to tariff shocks and geopolitical risks concentrated in single markets.
This diversification strategy strengthens resilience—an essential attribute in an era of supply-chain disruptions and shifting trade policies.
Foreign Investment and Investor Confidence
The pharmaceutical sector is among the top ten industries attracting foreign direct investment (FDI) in India. In the current financial year 2025–26 (up to September), foreign investment inflows in drugs and pharmaceuticals reached ₹13,193 crore. This steady inflow of capital signals international confidence in India’s manufacturing ecosystem, regulatory stability, and long-term growth prospects.
FDI not only augments production capacity but also facilitates technology transfer, research collaboration, and integration into global value chains. For rural and semi-urban manufacturing clusters, such investments translate into skilled employment and infrastructure development, linking local economies to global markets.
Trade Agreements and Market Access
India’s expanding network of trade agreements is further enhancing global market access for pharmaceuticals and medical devices.
The recently concluded Free Trade Agreement with the European Union significantly broadens opportunities in a market valued at approximately USD 572.3 billion. Tariff liberalisation under this agreement is expected to improve competitiveness for Indian pharmaceutical products and medical devices. Established manufacturing hubs in Maharashtra, Gujarat, Telangana, Karnataka, and Andhra Pradesh stand to benefit from export expansion, job creation, and MSME participation.
Similarly, the Comprehensive Economic and Trade Agreement signed with the United Kingdom in July 2025 provides zero-duty access for 56 pharmaceutical tariff lines. Given that the UK is India’s largest pharmaceutical export market in Europe, this measure is expected to enhance the competitiveness of Indian generics and reduce costs for manufacturers.
The Free Trade Agreement concluded with New Zealand in December 2025 grants zero-duty access across around 90 pharmaceutical tariff lines, deepening healthcare trade between the two nations.
Together, these agreements expand India’s trade footprint, integrate it more deeply into global healthcare supply chains, and reinforce its reputation as a dependable supplier of quality medicines.
Government-Led Manufacturing Push
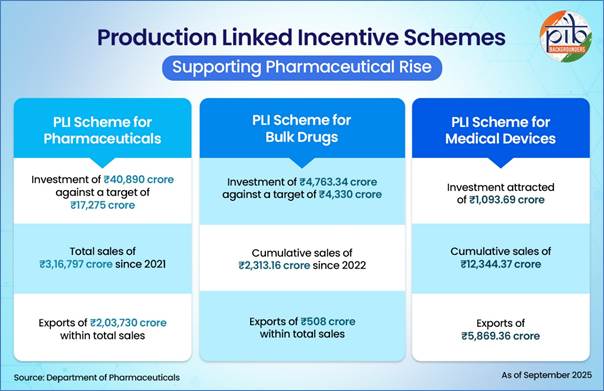
Central to India’s pharmaceutical rise are targeted policy interventions, particularly the Production Linked Incentive (PLI) schemes implemented by the Department of Pharmaceuticals.
The PLI Scheme for Pharmaceuticals supports domestic production of high-value biopharmaceuticals, complex generics, and specialty drugs. Since its launch in 2021, total sales under the scheme have reached ₹3.16 lakh crore, including exports worth ₹2.03 lakh crore. Investments of ₹40,890 crore—well above initial targets—have generated employment for nearly 97,000 individuals.
Complementing this is the PLI Scheme for Bulk Drugs, designed to reduce dependence on imported APIs, key starting materials (KSMs), and drug intermediates (DIs). Manufacturing capacity of over 55,100 metric tonnes per year has been established for 26 critical inputs, with cumulative sales exceeding ₹2,313 crore.
A separate PLI scheme for medical devices has catalysed investment and domestic production, with cumulative sales surpassing ₹12,344 crore.
Collectively, these initiatives have strengthened supply-chain resilience, reduced import dependency, and enhanced India’s manufacturing self-reliance.
Infrastructure Development: Parks and Clusters
To support ecosystem growth, the government has approved three bulk drug parks in Andhra Pradesh, Gujarat, and Himachal Pradesh, with total project costs exceeding ₹6,300 crore. These parks provide shared infrastructure—such as effluent treatment and testing facilities—reducing production costs and environmental risks.
Similarly, three medical device parks in Uttar Pradesh (Greater Noida), Madhya Pradesh (Ujjain), and Tamil Nadu (Kanchipuram) are at advanced stages of development. With nearly 200 manufacturers allotted land, these clusters are creating integrated manufacturing ecosystems that promote efficiency and innovation.
Innovation and Research: Moving Beyond Generics

While generics remain India’s strength, the future lies in innovation. The Scheme for Promotion of Research and Innovation in Pharma MedTech (PRIP) is steering the sector toward research-driven growth. Seven Centres of Excellence have been established at National Institutes of Pharmaceutical Education and Research (NIPERs), with 111 research projects approved and multiple patents filed.
In the Union Budget 2026–27, the government proposed the Biopharma SHAKTI initiative—an ambitious ₹10,000 crore programme aimed at positioning India as a global biopharmaceutical manufacturing hub. By upgrading existing NIPERs, establishing new institutions, and building a network of 1,000 accredited clinical trial sites, the initiative seeks to strengthen India’s capacity in biologics and biosimilars—complex, high-value therapies derived from living organisms.
Such measures mark a strategic transition from volume-driven generics to innovation-led growth.
Affordable Access at Home
While expanding exports, India has simultaneously prioritised affordability for its own citizens. The Pradhan Mantri Bhartiya Janaushadhi Pariyojana (PMBJP) operates over 18,600 Jan Aushadhi Kendras across the country, offering more than 2,100 medicines and 315 medical devices at significantly lower prices.
In 2024–25, sales under the scheme reached ₹2,022 crore, translating into estimated savings of ₹8,000 crore for citizens. Affordable sanitary napkins priced at ₹1 per pad have further advanced women’s health and dignity.
By reducing out-of-pocket expenditure, the scheme strengthens both public health and household financial stability.

Regulatory Architecture and Quality Assurance
India’s pharmaceutical credibility rests on a robust regulatory framework. The Central Drugs Standard Control Organisation (CDSCO) oversees drug approvals, clinical trials, and manufacturing standards under the Drugs and Cosmetics Act, 1940.
The National Pharmaceutical Pricing Authority (NPPA) ensures price control and availability of essential medicines under the Drugs (Prices Control) Order, 2013.
Meanwhile, the Indian Pharmacopoeia Commission (IPC) publishes official drug standards recognised in 19 countries, reinforcing international trust in Indian formulations.
This integrated regulatory ecosystem safeguards safety, affordability, and quality—pillars essential to sustaining export growth and domestic confidence.
Conclusion: A Forward-Looking Trajectory
India’s pharmaceutical journey reflects a rare convergence of public health commitment and economic ambition. From rural manufacturing units to global trade agreements, from generics to biologics, from affordability at home to export leadership abroad, the sector embodies resilience and adaptability.
Strong manufacturing capabilities, targeted policy support, regulatory credibility, expanding trade partnerships, and a growing innovation ecosystem collectively position India’s pharmaceuticals on a stable and forward-looking path. As global healthcare systems grapple with rising costs and inequitable access, India’s model—combining scale with affordability—offers a blueprint for inclusive growth.
In strengthening its pharmaceutical ecosystem, India is not merely expanding an industry; it is shaping a future where economic progress and public health reinforce one another, both at home and across the world.


Comments are closed.